Cancer
n., plural: cancers
ˈkænsər
Definition: The uncontrolled growth of cells in the body
Table of Contents
Cancer Definition
In biology, cancer is defined as the malignant growth due to uncontrolled cell division. It is now used as a general term for over a hundred diseases characterized by the uncontrolled, abnormal growth of cells. At advanced stages, the cells spread (metastasis) locally or through the bloodstream and lymphatic system to other parts of the body. The first historical description of this condition was in relation to breast carcinoma. The term “cancer” came from Ancient Greek καρκίνος (karkínos), meaning “crab”, because like a crab cancer cells seem to “grab on and won’t let go”. (Ref.1) Synonyms: neoplasia; growth; malignancy. Compare: tumor.
Cancer vs. Tumor
Cancers and tumors are similar in a way that both of these conditions are characterized by abnormal cell division that ends up in the formation of a mass of cells with no useful function. Tumors (also called neoplasms) are the more inclusive term for all abnormal cell growths. Thus, it can be said that cancer is a type of tumor. A cancerous tumor is one that has the potential to grow continuously and then spread to other tissues. Other hallmarks of cancers are as follows: new blood vessels form on the affected tissue, avoiding programmed cell death, and an unlimited number of cell divisions. (Ref.2)
What are the main causes of cancer?
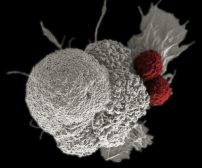
Cancers are not hereditary, meaning a cancer-stricken parent will not pass down cancer to the offspring. However, what is passed down is the predisposition to the condition. The offspring is at risk of developing cancer because of the inheritance of the defective genes from the parents. The defective gene can be due to a series or a set of mutations that made the gene abnormal. Oncogenes, for instance, are genes that were once normal genes (proto-oncogenes) that do their roles, e.g. in cell growth and proliferation, but have eventually transformed (mutated) into genes that can incite the normal cells to become cancerous. When these genes are present in the egg cell or sperm cell, they can be passed on to the next generation. As such, cancer due to the inherited mutated genes is what has been referred to as an inherited cancer. But as already mentioned, cancer itself is not hereditary but the abnormal gene that leads to cancer is. When oncogenes are activated, the once normal cells avoid apoptosis (programmed cell death) and proliferate instead.
Gene defects inherited from the parents account for only about 5% to 10% of all cancers. (Ref.3) This means that most cancers manifest from genes of an individual that have mutated eventually. In this case, the mutation is acquired later in life and therefore it does not come from the parent.
What are the things that make the genes mutate? Exposure to carcinogens is one of them. Carcinogens may be physical, chemical, or biological. They can damage the DNA. A problem arises when DNA damage involves the genes. The nucleotide sequence in the DNA may be disrupted resulting in a mutation. Not all mutations can lead to significant changes. However, when they do and the body fails to correct them by innate DNA repair mechanisms, the mutation will persist and can likely become heritable. When this happens, normal protein synthesis could be interrupted or the proteins can still be created but they will turn out to be dysfunctional. There are also instances wherein the genes are activated at a longer time than they should.
Chemical carcinogens include asbestos, tobacco smoke components, aflatoxin, and arsenic. (Ref.4) Some pathogens can transform a cell to become cancerous. Helicobacter pylori, hepatitis B or C virus, Epstein-Barr virus, HIV, and human papillomavirus can alter the genes of the host cell. (Ref. 4) These pathogens are, therefore, potential biological carcinogens.
Examples of physical carcinogens are electromagnetic radiation, e.g. UV, and ionizing radiation. Electromagnetic pollution (dirty electricity) has been apparent since the late 1800s. It is present everywhere –from utility power, switch-mode power energy supplies in electronics, variable speed motors in “smart appliances”, PV system inverters, and utility ground current. (Ref. 5)
Cancer is a result of the interaction between the genes and these carcinogens. However, other risk factors are also at play. Age is a crucial factor in the development of cancer. As the individual ages, the risk increases. The underlying reason is attributed to the gradual deterioration of the repair mechanism as the body ages. Aside from age, other risk factors are lifestyle (e.g. smoking, excessive drinking alcohol, lack of exercise, and poor diet) and exposure to environmental factors (e.g. pollution).
How does cancer start?
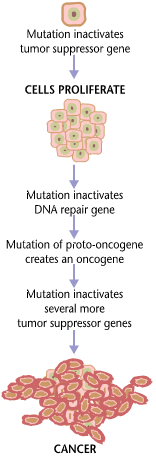
Cancer is presumed to be established as a result of several mutation occurrences. Multiple oncogenes are often involved, along with the mutated tumor suppressor genes. The biological role of the tumor suppressor gene is crucial in preventing tumors and cancers from forming. It regulates cell division, repairs errors in DNA, and incites cells to undergo apoptosis when it’s time. Thus, when the tumor suppressor genes mutate and are not corrected, they cannot function as they should. As a result, the cell will have no control over dividing. As the cells proliferate, a tumor that has the potential to become cancerous forms.
Are all cancers malignant?
Not all tumors are malignant. Tumors that aren’t malignant are called “benign”. Conversely, malignant tumors are cancers. Cancers, in turn, may be labeled by clinical stages based on the size and the extent of metastasis. Medical doctors conduct physical exams and tests, such as blood tests, imaging scans (e.g. MRI, CT scan, and ultrasound), and biopsy to determine the clinical stage of cancer.
The cancer stages are as follows: (Ref.6)
- Stage 0 (carcinoma in situ) – the presence of abnormal cells but not yet cancerous.
- Stage I (early-stage cancer) – cancer identified in small and one area.
- Stage II and III – cancer is relatively larger; some cancer cells have left the original location and have invaded nearby tissues or spread to lymph nodes.
- Stage IV (advanced or metastatic cancer) – cancer has spread to many other parts of the body.
How cancer works
In humans, there are about a hundred types of cancers. Some of these cancers include lung cancer, breast cancer, bone cancer, colorectal cancer, prostate cancer, skin cancer, brain cancer, and stomach cancer. All of them are caused by genetic changes that have accumulated and unrepaired. Oncogenes and mutated or inactivated tumor suppressor genes are genetic factors. The cell containing such faulty genes tends to divide uncontrollably; more so, it avoids death. As more and more of these cells containing abnormal genes divide, a lump grows on the site with no other function but to proliferate and soon invade other tissues. Some cells detach from the lump to spread to new locations in the body. This marks metastasis. Most fatalities from cancer are due to metastatic tumors. New tumors establish themselves well in various locations. They create new blood vessels, thereby gaining access to the nutrients in the bloodstream. Tumors with no specialized function whatsoever can impair the normal functioning of the organ from where they are located. In lung cancer, the presence of cancerous tumors in the lungs reduces the air capacity of the lungs. Thus, lung cancer patients suffer from difficulty in breathing. See the infographic on the right depicting cancer pathophysiology.
Note It!
Telomere — a Key to Cancer Cell’s Immortality
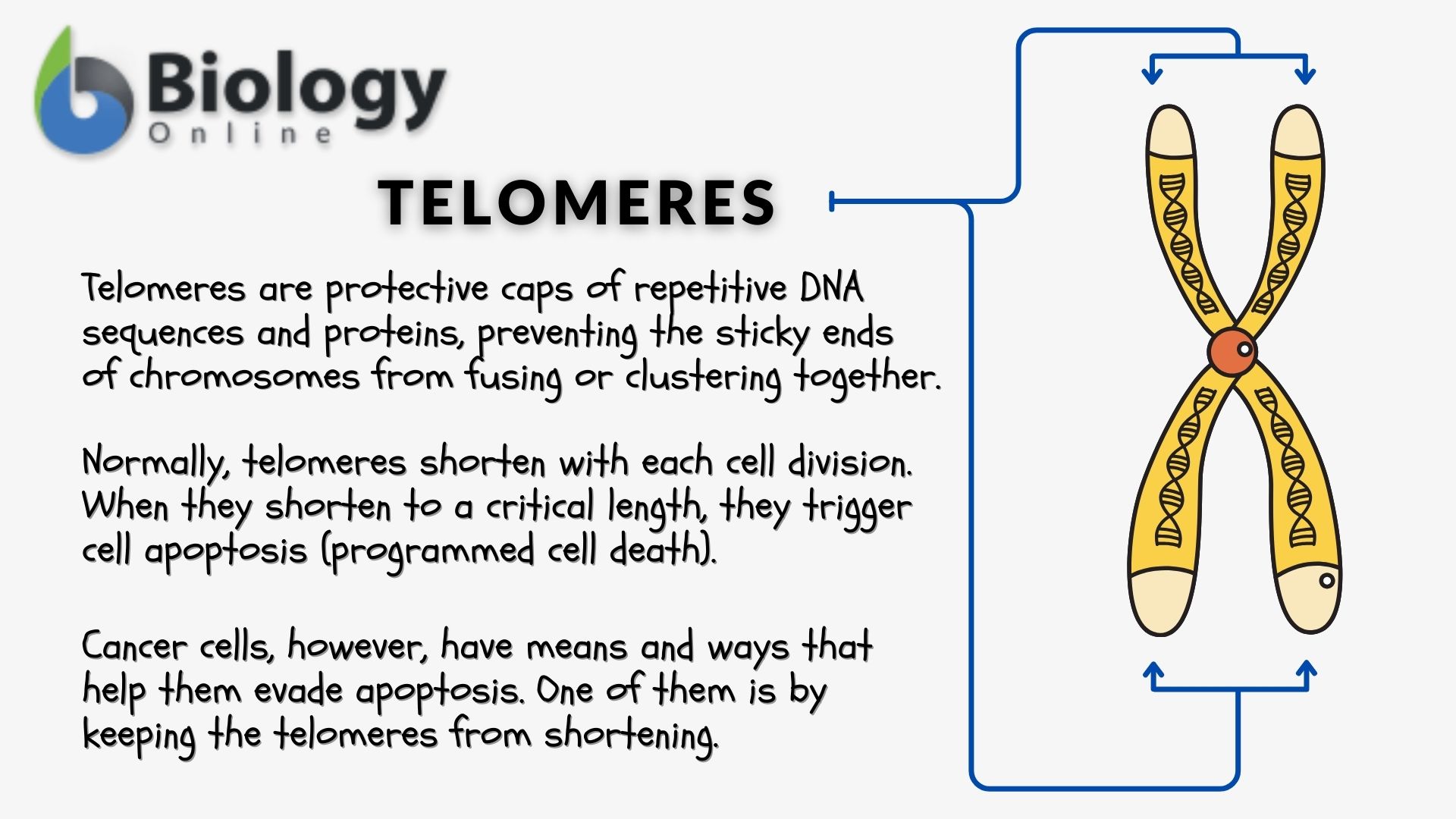
The telomeres — the terminal portions of the chromosomes — act as a ‘ cap‘ that prevents the sticky ends of the chromosomes from clumping together and protects chromosomal integrity, thus, maintaining genomic stability. But aside from that, they also play an essential role in cellular homeostasis. Their presence ensures that cells would not live and linger in the body much longer than they should.
Cells are naturally programmed to die through a process called apoptosis. This tightly regulated mechanism leads to the cell’s physiological death and subsequent removal from the body. The cell is incited to commit to the apoptotic pathway when it receives signals, which leads to a cascade of caspase activation. One primary driver of apoptosis is senescence or biological aging. At the cellular level, senescence refers to the stage in which the cell reaches the maximum number of cycles of cell division, and for that, it ceases to divide. The underlying reason is that the telomeres shorten gradually as a natural consequence of cell division. This form of senescence induced by the shortening of telomere with each round of cell division is replicative senescence. When the telomeres shorten at a critical point, they trigger a DNA damage response (DDR) signaling cascade, which when sustained promotes the transition from senescence to apoptosis. Phagocytes, such as macrophages, engulf and digest fragments and bodies of the cells that underwent apoptosis, thus, preventing them from accumulating.
In cells that have become cancerous, such a pathway is evaded. Many cancer cells enter alternative pathways that enable them to maintain telomere length, thus, allowing them to bypass replicative senescence. For instance, they upregulate the expression of telomerases, which are the enzymes that help preserve telomeres by replenishing them with repeat DNA sequences. Cancer cells may also carry out alternative lengthening of telomeres (ALT) mechanisms that help them evade apoptosis and the immune system. With these mechanisms, they can survive and remain viable, proliferating indefinitely and as such giving them a sense of “immortality” as they become cells that simply wouldn’t die.
Given the crucial role of telomerase in the immortality of cancer cells, it has served as a promising target for cancer treatment. Disrupting the expression of telomerase enzymes can make cancer cells susceptible to aging and death — an indication that cancer cells may be immortal but they are not entirely invincible.
See also
References
- What Are Tumors? (2018). Jhu.Edu. http://pathology.jhu.edu/pc/BasicTypes1.php
- Hanahan, D., & Weinberg, R. A. (2000). The Hallmarks of Cancer. Cell, 100(1), 57–70. https://doi.org/10.1016/s0092-8674(00)81683-9
- The American Cancer Society medical and editorial content team. (2014). Cancer.Org; American Cancer Society. https://www.cancer.org/cancer/cancer-causes/genetics/family-cancer-syndromes.html
- World Health Organization: WHO. (2018, September 12). Cancer. Who.Int; World Health Organization: WHO. https://www.who.int/en/news-room/fact-sheets/detail/cancer
- MEP (Dirty Electricity) Factsheet> MICRO-SURGE ELECTRIC POLLUTION. (2019, October 8). Building Biology Institute. https://buildingbiologyinstitute.org/free-fact-sheets/dirty-electricity/ (Dirty electricity as a cancer-causing agent)
- Langmaid, S. (2016, November 28). Stages of Cancer. WebMD; WebMD. https://www.webmd.com/cancer/cancer-stages#1
©BiologyOnline. Content provided and moderated by BiologyOnline Editors.