Table of Contents
Definition
noun
plural: galactoses
ga·lac·tose, ɡəˈlæktəʊz
An aldohexose monosaccharide (chemical formula: C6H12O6), similar to glucose structure (except that the orientations of H and OH on carbon 4 are exchanged), and when combined with glucose forms lactose
Details
Terminology
In 1856, the French biologist, Louis Pasteur 1822 –1895, was able to isolate galactose and called it lactose.1 The compound was later called galactose (or “glucose lactique”) as mentioned by Pierre Eugène Marcellin Berthelot 1827–1907, a French chemist, in his book, Chimie organique fondée sur la synthèse.2
Etymologically, galactose comes from the Ancient Greek γάλακτος (gálaktos, meaning “milk”) and -ose (denoting “sugar”).
Overview
Galactose is one of the three most common monosaccharides; the other two are glucose and fructose. Monosaccharides are the most fundamental type of carbohydrates. They are called simple sugars as opposed to the more complex forms such as oligosaccharides and polysaccharides. Monosaccharides can combine, though, to form complex carbohydrates via glycosidic bonds (glycosidic linkages).
Properties of galactose
Galactose is a hexose monosaccharide. It is an organic compound. Its general chemical formula is C6H12O6.The molar mass of galactose is 180.156 g/mol. The melting point is 168–170 °C. It is crystalline, water-soluble, and sweet tasting.
Galactose vs. Glucose vs. Fructose
Glucose, galactose, and fructose are the three most common natural monosaccharides. Nevertheless, glucose is the most abundant. The three have the same chemical formula: C6H12O6. Hence, they are a hexose-type of monosaccharide, owing to the six carbon atoms. Both glucose and galactose are aldoses whereas fructose is a ketose. Thus, glucose and galactose are more structurally alike. Nonetheless, glucose can be structurally identified from galactose based on the orientation of the hydroxyl group (OH) at carbon 4. Also, galactose has a higher melting point. Its melting point is 168–170 °C as opposed to glucose’s melting point of 146 °C. However, of the three, fructose has the lowest melting point (i.e. 103 °C).
Unlike glucose, galactose generally does not occur in free state. It usually is a constituent of complex biomolecules. For instance, galactose together with glucose forms lactose (milk sugar), which is a disaccharide. Thus, glucose is more often used than galactose or fructose in energy metabolism since it is more readily available. Without enough glucose, galactose enters glycolysis but galactose goes through initial steps to be converted into glucose 6-phosphate before it can proceed to glycolysis. The same principle happens to fructose; fructolysis (catabolism of fructose) entails fructose phosphorylation by fructokinase to produce fructose 1-phosphate, which is then cleaved by aldolase B into two trioses, dihydroxyacetone phosphate and glyceraldehyde.
Types of galactose
Two enantiomers of galactose exist: Dextrogalactose (D-galactose) and Levogalactose (L-galalactose). This nomenclature (based on Fischer projection) designates D– when the glucose stereoisomer rotates the plane polarized light in the clockwise direction. L– is when it rotates the plane polarized light in a counterclockwise direction. The dextrotatory form of galactose is obtained from milk sugar through hydrolysis. D-galactose is also present in sugar beets, seaweeds, and nerve cell membranes. Its levorotatory form is obtained from mucilages.
Common biological reactions involving galactose
Common biological reactions involving galactose
Through dehydration synthesis, a monosaccharide, such as galactose, binds to another monosaccharide with the release of water and the subsequent formation of a glycosidic bond. The joining of two monosaccharides produces a disaccharide whereas the joining of three to ten monosaccharide units forms an oligosaccharide. Polysaccharides are produced by the joining of multiple monosaccharides. In this regard, galactose joins with another monosaccharide to form a disaccharide. For instance, lactose is formed when galactose and glucose molecules are joined together. Another is the man-made disaccharide lactulose made up of galactose and fructose. As for polymers, a galactan is a polysaccharide consisting of repeating galactose units.
Common biological reactions involving galactose
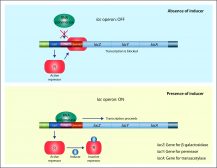
The process wherein complex carbohydrates are degraded into simpler forms, such as glucose and galactose, is called saccharification. It entails hydrolysis. In humans and other higher animals, this involves an enzymatic action. In a diet containing galactose (e.g. lactose in dairy products), digestion is aided by a β-galactosidase enzyme, lactase. Lactase catalyzes the hydrolysis of lactose and breaks β-glycosidic bond, resulting in the release of glucose and galactose in the small intestine. As for ceramide-containing diet, the lactase-glycosyleramidase complex breaks the β-glycosidic bond in glycolipids to release galactose.3 In the absence, or inadequacy, of lactase, lactose cannot be digested into simpler monosaccharides and as such, causes lactose intolerance. Lactose undigested in the small intestine moves to colon where the gut bacteria ferment it to lactic acid. As a result, methane and hydrogen gas are produced and cause discomfort, gut distention, and flatulence. Diarrhea ensues as water is drawn in to the intestine by the osmotically active lactic acid. Microorganisms, such as Escherichia coli, can metabolize lactose by producing β-galactosidase from its lac operon system.
Common biological reactions involving galactose
Galactose from dietary sources is taken up by the intestinal cells (enterocytes) through sodium-dependent glucose transporter, the same ATP-driven transport mechanism that absorbs glucose. Thus, glucose competes with galactose during intestinal absorption. Galactose leaves the intestinal cells and enters the bloodstream passively through glucose transporter- (GluT-) mediated transport.3
Common biological reactions involving galactose
Galactose is converted into glucose generally by a two-phase process. In the initial phase, β-D-galactose is converted into α-D-galactose by the enzyme mutarotase. In the last phase, α-D-galactose is converted into uridine diphosphate (UDP)-glucose. This last phase is often through the Leloir pathway. In this pathway, α-D-galactose is phosphorylated though galactokinase to produce galactose 1-phosphate. Next, galactose 1-phosphate acquires uridine monophosphate (UMP) group from UDP-glucose through the enzyme galactose-1-phosphate uridyltransferase to produce UDP-galactose. Then, UDP-galactose is interconverted into UDP-glucose by the enzyme UDP galactose-4′-epimerase. In humans and other organisms, an alternative to Leloir pathway is De Ley Duodoroff pathway. Galactose that is converted into glucose is the way by which galactose may enter the glycolytic pathway. The overall reaction would therefore be as follows:
Galactose + ATP → Glucose-1-phosphate + ADP + H+
Phosphoglucomutase catalyzes the isomerization of glucose 1-phosphate to glucose 6-phosphate. In humans, this galactose metabolism occurs in the liver.
Common biological reactions involving galactose
In humans and other mammals, some of the glucose molecules are converted into galactose so that there would more galactose to combine with glucose to produce lactose. This is especially important during milk production. The mammary gland secretes lactose as milk, especially during breastfeeding. N.B.: galactose may be obtained as well from dietary sources. The de novo synthesis of glucose and galactose in the mammary gland is called hexoneogenesis.
Common biological reactions involving galactose
Galactan is a polymer of galactose that occurs in hemicelluloses. In plants such as the axlewood ( Anogeissus latifolia) and acacia trees, galactose monomers link together and form galactans.
Common biological reactions involving galactose
Glycosylation is the process of adding a carbohydrate component, such as galactose, to certain proteins and lipids. Galactose forms part of certain glycolipids and glycoproteins. For instance, it may serve as a constituent of cerebroside (a glycolipid comprised of a carbohydrate and a sphingolipid). Cerebrosides are of two types: glucocerebrosides and galactocerebrosides, which have glucose and galactose carbohydrate residues, respectively.
Improper metabolism
Improper metabolism of galactose results in a condition called galactosemia. It is a rare metabolic disorder. One of the typical causes is the heritable genetic mutation involving the synthesis of an enzyme in the Leloir pathway, i.e. galactose 1-phosphate uridyl transferase. Galactosemics are not advised to consume galactose- (and lactose-) containing diet. Otherwise, it could result in diarrhea, vomiting, and eventually to cirrhosis.
Biological importance/functions
Galactose is one of the most common monosaccharides and plays various biological roles. For instance, it acts as an alternative to glucose when the latter is insufficient to the metabolic demands of an organism. It can enter glycolysis to synthesize energy. However, it must go through initial steps prior to entering the glycolytic pathway.
Galactose is a constituent of lactose, the disaccharide of milk. Humans and other milk-producing animals biosynthesize lactose from galactose and glucose. Milk is a vital source of nutrients, especially of neonates.
Galactose is a component of cerebrosides, and as such are called galactocerebrosides as opposed to glucocerebrosides containing glucose instead of galactose. Galactocerebrosides are commonly found in neural tissues and they are the main glycosphingolipid in the brain — presumably, the reason galactose is referred to as the brain sugar. Galactose that is later sulfated is referred to as sulfatide. Sulfatides play a role in immune response and nervous system signaling.
In plants, galactose occurs, such as in flaxseed mucilages and sugar beet. Galactan is a polymer of galactose found in hemicellulose in plants, such as the axlewood ( Anogeissus latifolia) and acacia trees.
Supplementary
Etymology
- Ancient Greek γάλακτος (gálaktos, meaning “milk”) + -ose (denoting “sugar”)
Abbreviation(s)
Chemical formula
- C6H12O6
Synonym(s)
Further reading
See also
- monosaccharide
- sugar
- lactose
- lactulose
- galactan
- glucose
- fructose
- Saccharification
- Leloir pathway
- Glycosylation
Reference
- Pasteur, L. (1856). “Note sur le sucre de lait” Note on milk sugar. Comptes rendus (in French). 42: 347–351. From page 348: Je propose de le nommer lactose. (I propose to name it lactose.)
- Berthelot, Marcellin. (1860). Chimie organique fondée sur la synthèse. Mallet-Bachelier (publisher). p.248. Retrieved January 24, 2019, from Link
- Carbohydrates: Galactose Metabolism Integrative Medical Biochemistry Examination and Board Review AccessPharmacy McGraw-Hill Medical. (2019, January 1). Retrieved from Link
© Biology Online. Content provided and moderated by Biology Online Editors