Substance
n., plural: substances
[ˈsʌbstənts]
Definition: any matter or material; a significant or vital component of a structure
Table of Contents
Substance Definition
What is a substance? The word substance finds varied usages in different science fields. In general science, a substance (noun) is a word used to refer to any type of matter or material. It can be used interchangeably with the word ‘material’. For example, in this sentence: “The tabletop is made up of a transparent glass substance”.
Here, the meaning of substance points to the material from which the tabletop is made up of. Let’s look at how the word is intended to be used in various science fields, such as biology and chemistry, with some examples and how one can define substance in different ways.
Substance Definition in Biology
In Biological Science, we use the term substance to denote the material or tissue of which a particular body part or organ is made up of.
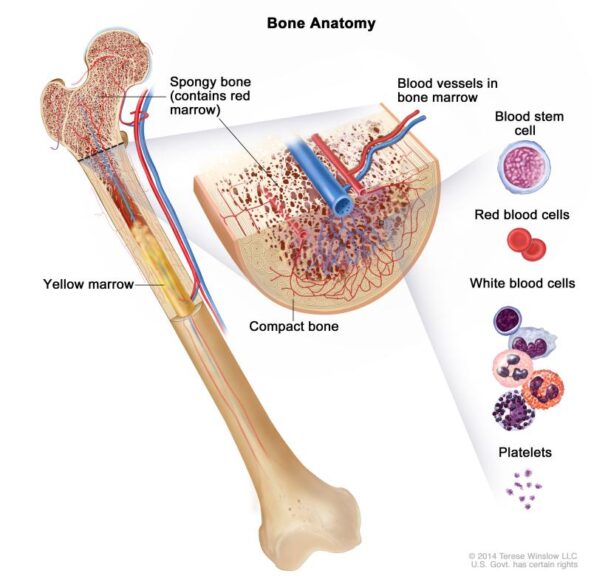
- Example: Bones have a spongy substance inside them called ‘bone marrow’ which is responsible for the production of bone marrow stem cells (cells that aid the production of more blood cells).
Substance Definition in Chemistry
The word substance is also used in the field of Chemistry to point to some “chemical substance” or any of the “chemicals” in general. Every chemical substance has a distinguished chemical composition, distinct essential nature, and characteristic properties.
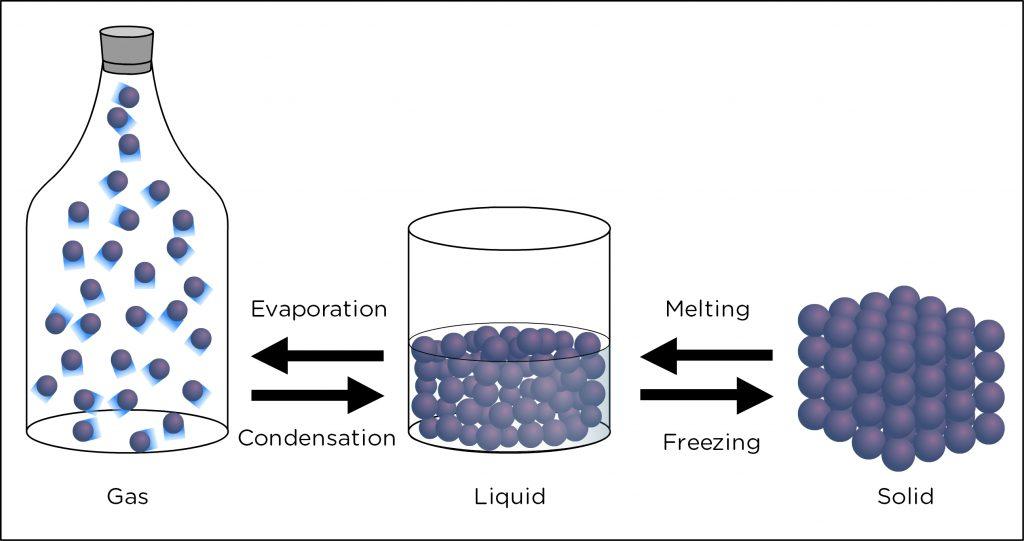
Chemical substances can’t be disintegrated into smaller parts as they are usually stable under constant surrounding conditions. So, simple physical separation processes aren’t enough to break them down. External pressure and temperature conditions that interfere with the stable chemical bonds of the chemical substance can only break them down or change the phase.
- Example: Water is a very stable chemical substance with definite chemical composition and distinct properties. Mere sieving the water can’t break it down. Only the application of heat or cold can change it into different phases like vapors or ice. And only by intervening with its chemical structure, by “oxidizing” it, that it can be disintegrated into oxygen gas and hydrogen ions and electrons. The equation for the oxidation of water molecules:
2H2O → O2 + 4H+ + 4e−
- Example: Water is a very stable chemical substance with definite chemical composition and distinct properties. Mere sieving the water can’t break it down. Only the application of heat or cold can change it into different phases like vapors or ice. And only by intervening with its chemical structure, by “oxidizing” it, that it can be disintegrated into oxygen gas and hydrogen ions and electrons. The equation for the oxidation of water molecules:
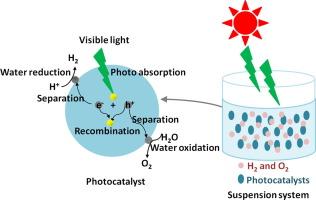
Substance Definition in Forensic Medicine
The term ‘substance’ is also used to denote illegal substances or drugs, as in substance abuse. In criminal justice cases and litigations, the word substance is associated with those chemical entities that have a chemical signature banned by the law. Possessing such substances or smuggling them can land you up in jail. These substances are usually addictive in nature, bad for your health, and thereby forbidden in civil societies by law.
- Example: There are a number of illegal substances like cocaine, LSD, psychedelic substances, scheduled drugs like heroin, methamphetamines, and some prescription substances, too, like marijuana, etc.

Substance in Scientific Works and Literature
In literature, a substance can be used to indicate ‘the most vital or essential part of something’.
- Example: “The substance of this study is the direct correlation of good diet and exercise with longevity and slowed down the aging process.” Usually, scientific research papers precisely state their most substantial results in the abstract of the paper.
Now that we have learned what substances mean, we can sprint ahead and learn the etymology of the word substance briefly.
Etymology of substance
The term ‘substance’ is a Middle English word. It has been derived from the Latin words ‘substantia’ meaning ‘being’, ‘essence’, ‘substant’, meaning ‘standing firm’. Originally, the first Latin word was ‘substare’.
[Note: Devoid of substance= substanceless]
Biology definition:
A biological substance refers to any biological molecule or biological material — from the tiniest molecules to the vital or significant components of biological tissues or organs, as well as the sample materials collected as specimens for biological testing and research.
- Examples of biochemical and biomolecular substances are elements, isotopes, ions, compounds, and other biological molecules
- Examples of vital biological components of organs or tissues are ground substance, spongy substance, etc.
- Examples of biological materials are tissues, bodily fluids, etc.
Etymology: from Latin “substantia”, literally meaning “standing under”.
Synonyms: particle; material; specimen.
Related forms: substantial (adjective)
Types of Substances
There are different types of substances based on various criteria but let’s focus on the types of substances we often apply in biology.
Molecular Substances
A molecule, in biology, refers to any small particle or substance, especially when they are associated with biological systems. Biomolecules are molecules that are produced or occur naturally in living organisms.
Examples of large biomolecules (macromolecules) are nucleic acids (DNA and RNA), proteins, carbohydrates, and lipids. Examples of small biological molecules are nucleotides, amino acids, monosaccharides, glycerol, and fatty acids. These are also referred to as micromolecules. However, there are also molecules that are rather chemical than biological in nature, such as water, minerals, and other inorganic compounds.
Defining molecules, in chemistry, is much stricter than it is in biology. Only those entities made up of an electrically neutral group of atoms (whether of the same kind or of different kinds of atoms) that can exist alone in a free state while the characteristic properties are retained can be molecules. Examples of chemical molecules are hydrogen molecules and water molecules.
Similarly, substances in chemistry (and biochemistry) are more strictly defined. A (pure) substance refers to any matter that has a definite set of characters and distinguished nature. In this regard, pure substances can be further classified as…
- an element (when made up of an atom or a set of the same atoms joined by a chemical bond forming a molecule). Examples: carbon, oxygen, hydrogen, diamond, and coal
- or a compound (when made up of more than one element joined by a chemical bond forming a molecule). Examples: carbon dioxide, water, salt
A ‘mixture’, as opposed to a pure substance, is made up of two or more pure substances that occur ‘together’ but are not held by a chemical bond, thus, can be separated by physical means such as distillation, crystallization, or filtration.
Although a mixture is basically made up of substances, it is not considered a substance because the components — although they blend — retain their own properties and the composition may vary.
For example, NaCl (salt) and H2O (water) are substances (compounds) that, together, form a salt solution. This solution is a mixture (homogeneous) and not a substance. A salt solution would have no definite property especially when the composition is altered by varying the amounts of the components (e.g, when more salt is subsequently added to the solution).
Light and heat are not substances as they are not forms of matter but forms of energy. In essence, they are not made up of atoms to confer distinct chemical properties.

Biological Materials
Biological materials or biological substances are those derived from or originated from an organism. Examples of human biological substances are human cells, tissues, organs, wastes, and bodily fluids such as blood, urine, semen, and other secretions.
For non-human biological materials, examples are biological products, cultures, genetically modified organisms, etc. These materials may be collected as specimens for biological testing or study.
Because biological materials may contain pathogens or any agents that pose health risks upon exposure, they are being classified based on hazard severity categories. These categories are used to classify biological substances for shipment. Examples of categorizing biological substances are as follows:
- Category A: a biological infectious material that may be life-threatening or may cause permanent disability or fatal disease to healthy humans or animals when exposed. Examples of pathogens contained in such materials are HIV and Hepatitis B viruses.
Example of shipping name: “Infectious Substance, Affecting Humans” - Category B: a biological material that contains or is suspected to contain pathogens or agents that are not life-threatening or incapable of causing permanent disability or fatal disease.
Example of shipping name: “Biological Substance, Category B” - Exempt Human and Animal specimens: biological specimens from a human or animal subject that have minimal likelihood of containing pathogens.
Example of shipping name: “Exempt Human Specimen” - Genetically Modified Organisms: biological materials that contain GMOs
Example of shipping name: “Genetically Modified Organisms”
If a biological specimen does not fall in any of these categories, such as substances with pathogens that have been inactivated, environmental samples, blood for transfusions, or tissues or organs for transplantations, they are regarded as Unregulated Biological Materials. (Environment, Health and Safety – University of North Carolina, 2014).
Watch this vid about shipping biological substance, Category B:
Endogenous vs. Exogenous substances
Substances may be classified based on their origin:
- Endogenous substances: substances obtained from or made available by the living organism. Thus, these substances originate from one’s own biological source or system. Examples: cells, tissues, organs
- Exogenous substances: substances obtained from or made available from an external source or outside the living organism or system. Thus, these substances would come from an outside source and not from one’s own biological source or system. Examples: pharmaceutical drugs, synthetic supplements
Substance Usage Examples
We have discussed many examples of substance in the definition section for clarity. Let’s look at some more examples of usage in biology and related fields:
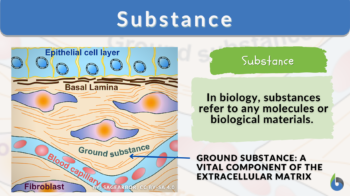
- Ground substance: Ground substance in different types of organisms like plants, algae, animal bodies, etc. They are amorphous and jelly-like in nature. They are usually present in the extracellular spaces (spaces between cells).

- Genetic substance: The primary genetic substance of the biological world is DNA (deoxyribonucleic acid). Although there is another genetic substance akin to DNA called RNA (ribonucleic acid).
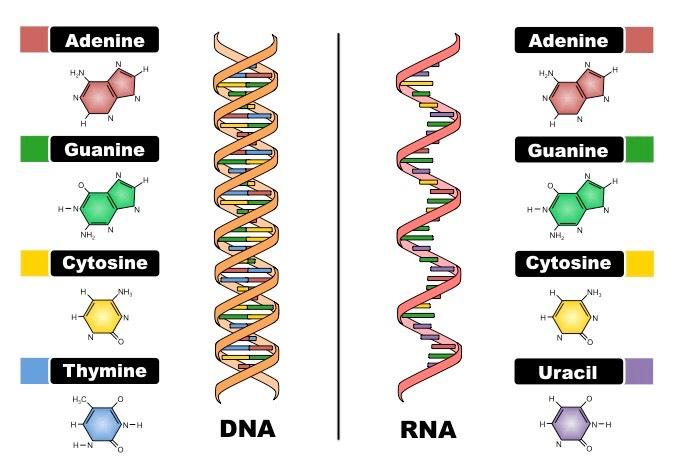
- Translucent substance: As opposed to opaque and transparent substances, translucent substances don’t block or allow the passage of light absolutely respectively. Rather translucent substances lie midway between these two substances and partially allow light to pass.

“Dangerous Substances”
According to a report by the Office on Drugs and Crime, United Nations, substance abuse ramped up during the pandemic. As the Covid-19 (coronavirus) hit the world in 2019, lockdowns, economic crashes, job losses and loss of loved ones left the world populations stricken with anxiety, depression, and a panic state.
The wiser way to deal with all this would have been to gather resources, calm the nerves and work consciously. But, at a global level, some people resorted to abusing substances like alcohol, cannabis, heroin, prescribed drugs, etc to cope with their anxieties. (Ref.4)
The important part of this conversation is that substance abuse can never help you cope with the problems of life. The cycle of substance abuse kicks off with dopamine, serotonin, and oxytocin rush, but it ends really bad.
What we need to learn here is that substances not just harm the user (consumer) but also the families associated and the people related to the user. Look at the bar chart representation of the different substances and the studied groups affected by them.
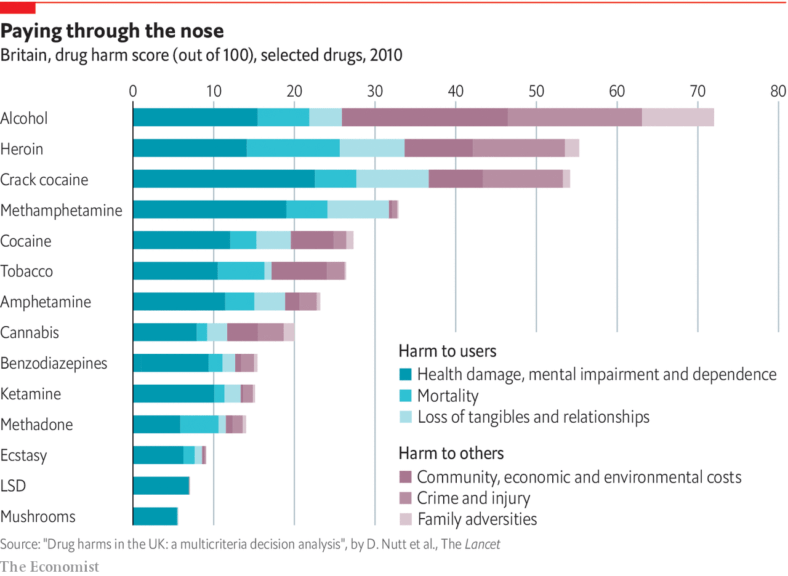
Answer the quiz below to check what you have learned so far about substances.
References
- Dan Kong, Yun Zheng, Marcin Kobielusz, Yiou Wang, Zhiming Bai, Wojciech Macyk, Xinchen Wang, Junwang Tang. (2018) Recent advances in visible light-driven water oxidation and reduction in suspension systems. Materials Today, 21(8): 897-924, ISSN 1369-7021, https://doi.org/10.1016/j.mattod.2018.04.009.
- PDQ Adult Treatment Editorial Board. Chronic Lymphocytic Leukemia Treatment (PDQ®): Patient Version. 2022 Mar 4. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National
- Cancer Institute (US); 2002-. [Figure, Anatomy of the bone. The…] Available from: https://www.ncbi.nlm.nih.gov/books/NBK66050/figure/CDR0000258005__182/
- Peckham M (2003). “Extracellular Matrix – ‘Ground substance'”. University of Leeds.
https://www.unodc.org/unodc/press/releases/2021/June/unodc-world-drug-report-2021_-pandemic-effects-ramp-up-drug-risks–as-youth-underestimate-cannabis-dangers.html - Environment, Health and Safety – University of North Carolina. (2014). Shipping Biological Materials Manual. https://ehs.unc.edu/wp-content/uploads/sites/229/2020/04/Shipping-Biological-Materials-Manual.pdf
©BiologyOnline.com. Content provided and moderated by Biology Online Editors.